文章信息
基金项目
- 国家自然科学基金面上项目(32172545); 海南省自然科学基金高层次人才项目(321RC636); 中央级公益性科研院所基本科研业务费专项“揭榜挂帅”项目(1630052022007); 国家现代农业(香蕉)产业技术体系品种改良岗位科学家项目(CARS-31-03); 热带作物生物育种全国重点实验室项目(NKLTCB202301)和中国热带农业科学院“热科优青”人才项目
作者简介
- 苗红霞,博士,中国热带农业科学院热带生物技术研究所研究员,香(大)蕉研究室副主任,主要从事果实品质形成与调控机理研究。主持国家自然科学基金面上项目和青年基金、国家公派留学基金等项目10余项,以第一或通信作者(含共同)在《Nature Plants》《Horticulture Research》《Journal of Integrative Plant Biology》等期刊发表SCI论文22篇; 获授权国家发明专利11件、新品种权1件; 获神农中华农业科技奖优秀创新团队奖1项、海南省自然科学奖一等奖1项; 入选首批海南省高层次人才、三亚崖州湾科技城D类人才和中国热带农业科学院“优青计划”; 兼任《Fruit Research》《广东农业科学》等期刊青年编委。苗红霞(1982—),女,博士,研究员,研究方向为果实品质形成与调控机理,E-mail:miaohongxia@itbb.org.cn.
通讯作者
- 刘菊华(1970—),女,博士,研究员,研究方向为香蕉遗传改良,E-mail:liujuhua@itbb.org.cn.
文章历史
- 收稿日期:2023-09-07
2. 中国热带农业科学院三亚研究院,海南 三亚 572025
2. Sanya Research Institute, Chinese Academy of Tropical Agricultural Sciences, Sanya 572025, China
淀粉是植物和藻类所特有的一种由葡萄糖组成的天然高分子聚合物[1],是食物中碳水化合物的主要来源,也是重要饲料组分和工业原料。全球每生产近20亿t粮食,其中有12亿~14亿t是淀粉[2]。淀粉以颗粒形式存在于质体,如光合组织的叶绿体或块茎、胚乳及块根等贮藏器官的淀粉体[3-4];且叶片中合成的淀粉称为暂时性淀粉,种子、果实或块茎中合成的淀粉称为贮藏性淀粉[5]。根据人体对淀粉消化吸收方式不同,分为可消化淀粉、慢消化淀粉和抗消化淀粉[6-7],其中抗消化淀粉又称为抗性淀粉(Resistant starch, RS)。RS由英国生理学家Englyst于1982年在体外环境对非淀粉多糖进行酶解实验中首次发现并命名[8],是指120 min内不能被健康人体小肠所消化和吸收、但却能在大肠中发酵的淀粉及其淀粉降解物的总称[9]。依据其化学结构、来源和性质不同,RS共分为5种类型:RS1(物理包埋淀粉,Physically trapped starch)[7, 10-11]、RS2(抗性颗粒淀粉,Resistant starch granules)[12-13]、RS3(回生淀粉,Retrograded starch)[14-15]、RS4(化学改性淀粉,Chemical modified starch)[14, 16-20]和RS5(直链淀粉- 脂质复合物,Amylose-lipid complexes)[21-24]。
香蕉(Musa spp.)是世界第四大粮食作物,约有135个国家或地区种植,是全球近20亿人口碳水化合物来源[25]。香蕉是含天然RS最高的作物之一,采收期果实中RS含量为40%~50%[26],远高于水稻(< 3%)、小麦(<3.5%)、高直连淀粉玉米(<22.4%)和大麦(<15%)等作物[26-28]。鉴于此,一方面,香蕉可作为功能性食物的直接来源,也可作为健康食品研发的原材料;另一方面,香蕉是研究高RS形成分子机制的理想材料,且香蕉A和B全基因组测序完成也为其研究提供了大数据平台。本文综述了1982年以来在香蕉RS的类型及其消化特性、淀粉颗粒形态特征及其影响因素、RS积累和降解特点及其对果实品质形成的影响、RS合成相关基因研究、香蕉RS制备方法及其在食品加工应用等方面取得的进展,以期为香蕉RS前沿基础研究、开发和综合利用奠定基础。
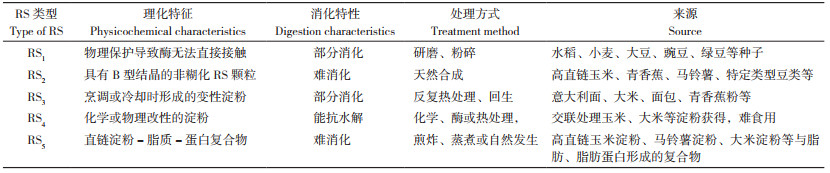
1 香蕉抗性淀粉类型及其消化特性RS包括RS1~RS5 5种类型,且不同类型RS理化特征存在较大差异,导致其消化特性、用途或应用领域存在明显不同,为个性化设计不同RS类型、精准调控糖脂代谢和维护人体健康提供依据。(1)RS1具有完整的细胞壁结构[10],可通过充分研磨和咀嚼来促进RS1被人体小肠消化吸收,从而被人体食用;(2)RS2属于天然淀粉颗粒,具有特殊结晶结构,可直接鲜食(如香蕉),避免加工过程中对RS2结构破坏[12-13];(3)RS3因其颗粒结构紧密、热稳定型性好等特点,在热加工食品中能够保持稳定,可作为添加剂和膳食纤维添加至油炸食品、面包等食品中,从而改善食品脆性、口感等品质,在食品工业中具有广泛应用前景[18-19];(4)RS4是通过与化学试剂交联、醚化或酯化反应引入新的化学官能团至淀粉分子内部,使分子结构发生改变而产生酶抗性的改性淀粉[14, 16];(5)RS5是在外界条件作用下,直链淀粉形成左手螺旋空腔结构、脂质进入直链淀粉的螺旋空腔发生不同程度的耦合,形成的一种复杂直链淀粉- 脂质复合物结构[21],也可以是含有少量或不含直链淀粉的淀粉(蜡质淀粉又称支链淀粉)与脂质形成的复合物[24, 29]。
香蕉RS属于RS2类型。RS2是唯一被美国食品和药物管理局确定为膳食纤维的RS。RS2对淀粉酶活性的抗性取决于淀粉颗粒结晶结构(包括A、B、C型),其中B型热稳定性好、抗消化能力最强[7, 10-11]。香蕉RS2作为一种功能性膳食纤维配料添加至意大利面条、面包、饼干等食品中,可以不改变食物的物理、化学性质,还具有降低血糖指数、增加饱腹感等保健功能,特别适用于糖尿病患者等特殊人群食用[12-13]。有关RS1~RS5理化特征、消化特性、处理方式及来源等介绍,详见表 1。
2 香蕉淀粉颗粒形态特征及其影响因素
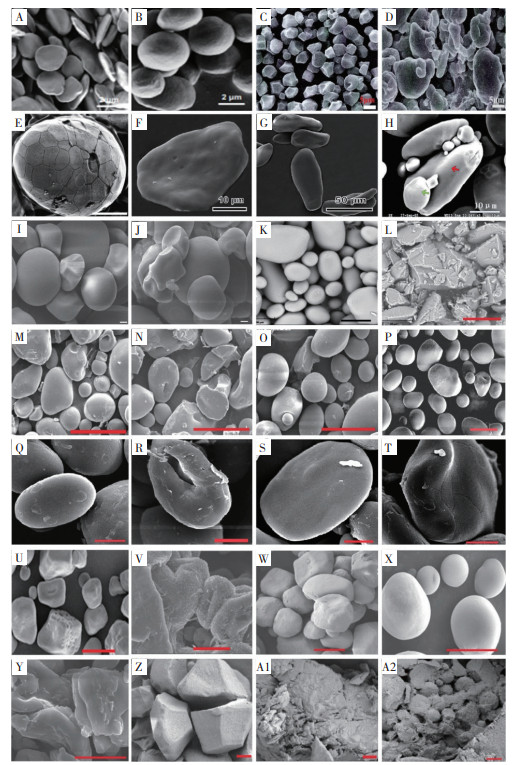
淀粉以颗粒形式贮存于植物根、球茎、块茎、种子和果肉(如香蕉等)中。不同类型淀粉颗粒形态、大小在物种间和品种间存在较大差异。在作物遗传育种中,淀粉形态、粒度、颗粒表面纹理等特征可作为种质创新、品种鉴别、突变体鉴定和优异变异性状筛选的重要参考。根据扫描电镜(Scanning Electron Microscope,SEM)观察,自然界天然淀粉颗粒形态主要有以下3种:(1)复合颗粒结构,由紧密排列的多边形颗粒组成,类似球形结构,具有膜质的隔膜包裹(图 1C、D、E),如水稻、高直链玉米等淀粉颗粒;(2)离散颗粒结构,直径为2~30 μm,淀粉颗粒呈多面体、球体、椭圆形、圆盘形等形态(图 1A、B、F、G),如香蕉、绿豆等淀粉颗粒;(3)双峰颗粒结构,通常由小的球形(B型)和较大的晶状体(A型)两种颗粒构成,如小麦、马铃薯等淀粉颗粒(图 1H、K)[30]。
|
| A:野生型拟南芥淀粉颗粒[40];B:dpe2-1/SS4-2双突变体拟南芥淀粉颗粒[4];C:水稻淀粉颗粒[31];D:高直链水稻淀粉颗粒[31];E:芦苇胚乳淀粉颗粒[40];F:Mysore香蕉淀粉颗粒[33];G:Figo大蕉淀粉颗粒[33];H:小麦A型和B型淀粉颗粒,红色箭头表示A型,绿色箭头表示B型[41];I:甘薯淀粉颗粒(Bar=2 μm)[17];J:甘薯RS3颗粒(Bar=2 μm)[17];K:马铃薯淀粉颗粒(Bar=30 μm)[36];L:微波- 双酶法制备的马铃薯RS3颗粒(Bar=30 μm)[36];M:湿热处理的马铃薯RS3颗粒(Bar=50 μm)[37];N:乳酸和湿热处理的马铃薯RS4颗粒(Bar=50 μm)[37];O:柠檬酸- 湿热处理的马铃薯淀粉颗粒(Bar=50 μm)[37];P:OSA处理的马铃薯RS4颗粒(Bar=50 μm)[38];Q:小扁豆淀粉颗粒(Bar=7 μm)[39];R:OSA处理的小扁豆RS4颗粒(Bar=7 μm)[39];S:香蕉淀粉颗粒(Bar=7 μm)[39];T:OSA处理的香蕉RS4颗粒(Bar=7 μm)[39];U:玉米淀粉颗粒(Bar=10 μm)[42];V:醚化交联制备的玉米RS4颗粒(Bar=10 μm)[42];W:玉米淀粉- 原花青素复合物RS5颗粒[43] (Bar=10 μm);X:慈姑淀粉颗粒(Bar=10 μm)[44];Y:超声波制备的慈姑淀粉- 亚油酸复合物RS5颗粒[44](Bar=10 μm);Z:水稻淀粉- 没食子酸复合物RS5颗粒(Bar=1 μm)[45];A1:水稻淀粉- 介子酸复合物RS5颗粒(Bar=1 μm)[45];A2:超声波处理和水热制备的水稻- 多酚复合物RS5颗粒(Bar=1 μm)[45] A: Starch granules of wild-type Arabidopsis[40]; B: Starch granules of dpe2-1/SS4-2 double mutants in Arabidopsis[4]; C: Starch granules of rice[31]; D: Starch granules of high-amylose rice[31]; E: Endosperm starch granules of reed[40]; F: Starch granules of Mysore banana variety[33]; G: Starch granules of Figo banana variety[33]; H: Wheat A- and B-type starch granules, with red arrow indicating A-type and green arrows indicating B-type granules[41]; I: Starch granules of sweet potato (Bar=2 μm)[17]; J: Resistant starch 3 (RS3) granules of sweet potato (Bar=2 μm)[17]; K: Starch granules of potato (Bar=30 μm)[36]; L: Potato RS3 granules prepared by microwave-double enzyme method (Bar=30 μm)[36]; M: Potato RS3 granules with moist heat treatment (Bar=50 μm)[37]; N: Potato resistant starch 4 (RS4) granules with lactic acid and moist heat treatment (Bar=50 μm)[37]; O: Potato RS4 granules with citric acid and moist heat treatment (Bar=50 μm)[37]; P: Potato RS4 granules with Octenyl Succinic Anhydride (OSA) treatment (Bar=50 μm)[38]; Q: Starch granules of lentil (Bar=7 μm)[39]; R: Lentil RS4 granules with OSA treatment (Bar=7 μm)[39]; S: Starch granules of banana (Bar=7 μm)[39]; T: Banana RS4 granules with OSA treatment (Bar=7 μm)[39]; U: Starch granules of maize (Bar=10 μm)[42]; V: Maize RS4 granules prepared by etherification crosslinking way (Bar=10 μm)[42]; W: Resistant starch 5 (RS5) granules composed of maize starch-procyanidin complex (Bar=10 μm)[43]; X: Starch granules of arrowhead (Bar=10 μm)[44]; Y: RS5 granules composed of arrowhead starch-linoleic acid complex with ultrasound treatment (Bar=10 μm)[44]; Z: RS5 granules composed of rice starch-gallic acid complex (Bar=1 μm)[45]; A1: RS5 granules of rice starch-mesonic acid complex (Bar=1 μm)[45]; A2: RS5 granules of rice starch-polyphenol complex with ultrasomic and hydrothermal treatment (Bar=1 μm)[45] 图 1 不同植物淀粉、RS颗粒扫描电镜 Fig. 1 Scanning electron micrographs of starch and RS granules from different plant species |
2.1 香蕉淀粉颗粒形态特征
香蕉淀粉颗粒形态、大小与拟南芥和水稻等模式植物明显不同。拟南芥野生型叶片淀粉呈圆盘状,形状扁平且体积较小(图 1A),其dpe2-1/SS4-2双突变体(图 1B)和dpe2-1/phs1a/ss4-2三突变体淀粉颗粒多呈圆球形、颗粒变大[4]。普通水稻淀粉颗粒为多边形,表面光滑且棱角分明(图 1C),其高直链淀粉突变体的胚乳淀粉呈椭圆形的大淀粉颗粒和较小的细长淀粉颗粒[31]。同时,禾本科种子胚乳中淀粉粒包括复合、简单和双峰等3种颗粒结构[32]。然而,香蕉淀粉颗粒多呈圆形、不规则三角形和棒形,颗粒大小为7~60 μm,且不同香蕉品种的淀粉颗粒存在明显差异,如栽培香蕉‘Mysore’品种青香蕉淀粉颗粒呈多边形,扁平状颗粒;而‘Figo’品种青大蕉淀粉颗粒呈棒状(图 1F、G)[33];‘巴西蕉’淀粉颗粒为不规则三角形,‘宝岛蕉’淀粉颗粒呈圆形,而‘红香蕉’淀粉颗粒多为棒形[34]。目前关于香蕉淀粉颗粒形态特征已有较多报道,但对天然存在的香蕉RS2颗粒形态特征及超微结构研究报道较少。有研究表明,香蕉RS2颗粒呈表面不规则椭圆形,且推测其颗粒表面有一层光滑的致密层(厚约几微米),是阻碍酶与淀粉接触、抑制淀粉降解的重要因素[35],但RS2颗粒表面致密层的结构物质仍不清楚。此外,由于香蕉RS2颗粒不耐高温,导致其在食品加工应用方面有一定局限性,所以部分学者利用不同加工处理条件或络合反应将其改性为RS3或RS4类型,拓展其在食品加工、医药等领域的广泛应用。
2.2 香蕉淀粉颗粒形态特征变化的影响因素2.2.1 加工处理条件对香蕉淀粉颗粒形态特征变化的影响 在加工过程中,RS颗粒形态改变是判断改性淀粉的一个重要指标。酸/碱处理及湿热处理组合会导致RS3和RS4颗粒形态及其结构发生改变。但与甘薯、马铃薯、水稻、玉米RS3或RS4颗粒形态相比较,香蕉RS4颗粒呈现与水稻、马铃薯RS4颗粒相似表型,但与甘薯、玉米RS4颗粒形态不同。甘薯天然淀粉呈光滑的卵圆形或不规则形状(图 1I),然而经过高温湿热和强碱等处理后,甘薯RS3颗粒表面呈现不同程度的凹陷及碎片(图 1J)[17]。马铃薯原淀粉颗粒呈不规则椭圆形,表面比较光滑(图 1K);而经过微波- 酶解法制备的马铃薯RS3颗粒呈不规则的块状结构,且表面粗糙、呈沟壑状层状起伏纹理,同时伴有颗粒碎屑[36](图 1L)。另有研究发现,湿热处理的马铃薯淀粉表面糊化并开裂,在一些颗粒表面出现盘状凹陷(图 1M)[37];不同酸处理对马铃薯淀粉颗粒结构的破坏程度存在差异,柠檬酸- 湿热处理的马铃薯淀粉颗粒则保持完整形态(图 1O)[37];而经过乳酸- 湿热处理后马铃薯淀粉完整性被破坏,淀粉被分解成更小的小块[37](图 1N)。淀粉在微碱性条件下,淀粉颗粒可与辛烯基琥珀酸(Octenyl succinic anhydride, OSA)发生酯化反应生成OSA淀粉;OSA淀粉属于RS4,具有良好的乳化性能和增稠特性,常作为新型食品添加剂。马铃薯原淀粉颗粒表面比较光滑,而经过OSA处理的马铃薯淀粉颗粒表面略显粗糙,边缘清晰度降低,侧面呈现多孔结构,同时出现许多空腔[38](图 1P)。小扁豆原淀粉多为椭圆形、部分为不规则球形(图 1Q),经过OSA酯化后淀粉颗粒表面出现一些空腔(图 1R)。然而,香蕉淀粉颗粒多呈圆形(图 1S),酯化香蕉RS4颗粒表面则出现一些裂纹(图 1T),这一特征与经过OSA酯化的早籼稻、马铃薯RS4颗粒表面有裂纹表型相似[39],与醚化、交联处理后玉米RS4颗粒表面粗糙且出现较多点状突起形态不同(图 1V)。
2.2.2 络合反应对香蕉淀粉颗粒形态特征变化的影响 众所周知,淀粉颗粒可与醇类、脂类、酚类等物质形成络合复合物,且具有抗血糖作用,因为它们可以抑制多种消化酶(如胰蛋白酶)。慈姑淀粉颗粒多呈球形、表面光滑(图 1X),通过超声波辅助制备的慈姑淀粉- 油脂复合物中淀粉颗粒形态被破坏,淀粉- 亚油酸络合物形态增大,颗粒具有表面光滑的海绵状结构,呈RS5颗粒(图 1Y)。同时,有研究显示,玉米、马铃薯来源的淀粉可与从高粱中提取的多酚类物质原花青素形成半结晶(Ⅱ)内螺旋V型复合物,该复合物很大程度上对淀粉酶和淀粉葡糖苷酶水解具有抗性,在酶解2 h后仍能保持完整RS5颗粒形态(图 1W)。水稻淀粉颗粒呈多边形、形状不规则且表面光滑,其与没食子酸经过水热法反应形成的淀粉- 酚类复合物淀粉颗粒形态变化不大,只是表面稍微粗糙(图 1Z)。使用超声波和水热处理制备的淀粉- 介子酸多酚复合物淀粉的天然结构被破坏,此时通常形成较多片层和凹槽的多孔结构[45](图 1A1),水热和超声处理的淀粉-酚类复合物由于超声波空化现象易形成破碎颗粒[45](图 1A2)。然而,络合反应对香蕉淀粉颗粒形态变化的影响未见报道,仅有应用米糠油、椰子油、葵花油制备香蕉淀粉- 脂质复合物的研究[46]。这些淀粉颗粒与醇类、脂类、酚类物质等形成的络合物,将为改性淀粉应用于人类健康开辟了新途径。
3 香蕉抗性淀粉积累特征及其降解对果实品质形成的影响 3.1 香蕉抗性淀粉积累特征香蕉RS积累动态变化规律与谷类作物[26-28]不同。在玉米、小麦、水稻、高粱等作物中,籽粒在花后不久开始合成淀粉,总淀粉积累多呈“S”型曲线增长,总体表现为“慢- 快- 慢”的增长趋势;在籽粒成熟期,其总淀粉含量达到最高,直链淀粉和RS含量也达到峰值[29-30, 47-49]。香蕉果实中总淀粉、直链淀粉和RS等在抽蕾后20 d开始大量合成,抽蕾后30 d均呈现直线增加趋势,至抽蕾后70 d时达到最大值;抽蕾70 d后RS含量开始下降(图 2A)。相关性分析结果表明,香蕉果实生长发育过程中,其RS合成与直链淀粉含量变化呈显著正相关,与总淀粉和支链淀粉含量相关性不显著(图 2A)[26]。

|
| A:香蕉采前总淀粉、直链淀粉、支链淀粉与抗性淀粉含量变化;B:采后不同处理香蕉抗性淀粉含量变化;C:自然后熟香蕉可溶性糖含量变化;D:采后乙烯处理香蕉可溶性糖含量变化;E:采后1-MCP处理香蕉可溶性糖含量变化 A: Changes in contents of total starch, amylose, amylopectin and resistant starch (RS) in banana fruit before harvest; B: Changes in RS content in banana fruit after harvest with different treatments; C: Changes in soluble sugar content in banana fruit after harvest; D: Changes in soluble sugar content in banana fruit treated with ethylene after harvest; E: Changes in soluble sugar content in banana fruit treated with 1-MCP after harvest 图 2 香蕉果实采前和采后总淀粉、直链淀粉、支链淀粉、抗性淀粉与可溶性糖含量变化[26, 50] Fig. 2 Changes in contents of total starch, amylose, amylopectin, resistant starch and soluble sugar in banana fruit before and after harvest[26, 50] |
3.2 香蕉抗性淀粉降解对果实品质形成的影响
香蕉属于典型的呼吸跃变型和淀粉转化型果实[51-52],采后随着果实成熟发生了复杂的生理生化变化,RS快速降解,至可食期RS含量仅为4.2%[26]。香蕉果实淀粉降解涉及淀粉降解酶基因表达和转录因子调控的生物学过程。已有研究表明,α-淀粉酶(α-amylase, AMY)、β-淀粉酶(β-amylase, BAM)和异淀粉酶(Isoamylase, ISA)等参与香蕉果实淀粉降解[53-54],且转录因子MabHLH6通过与11个淀粉降解关键酶基因的启动子结合调控果实淀粉降解过程[55],但以上研究均未涉及香蕉RS降解。苗红霞等[26]发现,香蕉RS含量在采后第2 d急剧下降,而此时总淀粉、直链淀粉、支链淀粉含量的变化并不明显,认为这一时期香蕉并未发生明显的淀粉降解,而主要是RS结构变化,直链淀粉和支链淀粉从致密层结构中暴露出来,增加了各种淀粉酶的可及性,据此推测淀粉粒表面致密层结构变化是RS降解的关键,发生在香蕉果实采后早期。
自然后熟过程中,香蕉RS含量迅速下降(图 2B),而可溶性糖(蔗糖、果糖、葡萄糖)含量呈先增加后减少的单峰型变化(图 2C);乙烯处理的香蕉RS和可溶性糖(蔗糖、果糖、葡萄糖)含量变化趋势与自然后熟过程趋势一致(图 2C、D);而1-MCP处理后果实RS含量呈先增加后减少的单峰型变化,蔗糖、果糖、葡萄糖含量呈逐渐上升趋势(图 2B、E)。相关性分析发现,采后成熟过程中,香蕉果实RS含量变化与可溶性糖(蔗糖、果糖、葡萄糖)含量均呈显著或极显著相关性[26, 50],表明RS降解对香蕉果实甜味物质形成具有重要作用。
4 香蕉抗性淀粉合成相关基因研究进展淀粉合成是一个复杂的生化过程,主要涉及5种关键酶,包括腺苷二磷酸葡萄糖焦磷酸化酶(ADP-glucose pyrophosphorylase, AGPase)、颗粒结合淀粉合成酶(Granule-bound starch synthase, GBSS)、可溶性淀粉合成酶(Soluble starch synthase, SSS)、淀粉分支酶(Starch branching enzyme, SBE)和淀粉去分支酶(Starch debranching enzyme, DBE)[56]。而GBSS的作用是通过α-1, 4-D-糖苷键将腺苷二磷酸葡萄糖(Adenosine diphosphateglucose, ADPG)中的葡萄糖残基添加到葡聚糖非还原端,延长葡聚糖的直链。此外,GBSS还能与植物发育过程中的淀粉颗粒紧密结合,是淀粉合成酶中唯一有活力的蛋白[57]。单子叶植物中的GBSS主要有GBSSI和GBSSII两种类型[58]:GBSSII主要控制根、茎、叶等营养器官直链淀粉合成,双子叶植物只有GBSSII单个家族,且功能与单子叶植物GBSSII相似;而GBSSI由waxy基因编码,主要控制种子、胚乳、果实等贮藏器官直链淀粉合成[58]。
4.1 GBSSI表达直接或间接参与抗性淀粉合成人们曾普遍认为GBSSI表达主要控制种子、胚乳、果实等贮藏器官直链淀粉合成,但越来越多的新证据表明GBSSI表达直接或间接参与RS合成。Zhou等[59]研究发现,GBSSI和SSIIIa共同参与水稻RS合成,且SSIIIa对RS合成的调控依赖于GBSSI基因的高表达,进而在ssIIIa突变体背景下降低GBSSI表达、导致胚乳中RS含量下降。随后,在粳稻突变株be2中过表达籼稻GBSSI基因发现,其转基因株系子代胚乳中RS含量明显高于亲本[60]。而水稻γ278突变体籽粒中RS含量显著增加的原因,主要是由于GBSSI、SSIIa和SSIIIa 3个基因的序列突变及其表达特性发生改变;进一步通过野生型×突变体杂交获得F2群体验证突变组合的作用,最终确定GBSSI: ssIIa: ssIIIa组合决定了F2群体高RS含量的合成[61]。Zhang等[62]将104个重测序水稻品系的RS含量与2 288 867个位点的Single Nucleotide Polymorphisms(SNP)数据集进行关联分析,发现其中一个SNP关联到所有重测序品系的RS含量变化,被直接定位在GBSSI基因上;另一个SNP是SBEIIa基因仅关联到INDICA品系的RS含量变化,表明GBSSI基因影响RS含量变化在水稻重测序品系中存在普遍性。在小麦中也有报道认为,突变体株系中RS含量明显提升的原因是GBSSI及其同源基因的显著高表达[63]。
4.2 转录因子调控GBSSI表达影响抗性淀粉含量研究表明,一些转录因子如Basic leucine zipper(bZIP)、Nuclear factor-Y(NF-Y)、Myelocytomatosis(MYC)和MADS-box蛋白,可以促进或抑制淀粉合成相关基因的表达,直接影响RS合成[64]。在水稻中,OsbZIP33可与GBSSI和SBEI基因启动子区域ACGT7motif结合,从而促进其上调表达[65]。OsbZIP58结合one finger(Dof)家族转录因子Rice prolamin box binding factor(RPBF)可以促进OsAGPL3、GBSSI、SSIIa、SBEI、SBEIIb和DBE2表达;在水稻osbzip58突变体中,总淀粉、直链淀粉和脂质含量显著降低、支链淀粉短链减少、支链淀粉长链显著增加[66-67]。Dehydration-response element-binding(DREB)转录因子SALT-RESPONSIVE ERF1(SERF1)可与GBSSI和RPBF启动子区结合,从而抑制其表达;在serf1突变体中,总淀粉和氨基酸含量明显增加[68]。MYC家族成员OsBP-5与GBSSI启动子区CAACGTG motif结合,且其与AP2/EREBP家族成员OsEBP-89互作从而激活GBSSI转录[69];在水稻OsBP-5 RNAi干扰株系中,直链淀粉含量明显下降[69]。NF-YB1与NF-YC12、bHLH144相互作用形成三元复合体,且与GBSSI启动子区G-box结合直接激活其表达;在NF-YB1、NF-YC12或bHLH144突变体中,总淀粉、直链淀粉和油脂含量显著减少,但水稻胚乳中蛋白含量明显增加[70]。Feng等[71]研究表明,MADS-box家族成员OsMADS14与NF-YB1相互作用并直接结合OsAGPL2和GBSSI启动子区CArG盒促进其转录;在OsMADS14突变体中,淀粉颗粒的形状和大小明显改变,总淀粉和直链淀粉含量显著减少,可溶性糖含量增加。综上所述,这些转录因子可能形成一个复杂的调控网络来调节GBSSI表达,从而影响直链淀粉合成或淀粉精细结构,最终影响RS含量。
4.3 GBSSI是促进香蕉果实抗性淀粉合成的关键基因为探索GBSSI如何作用于香蕉果实RS合成,苗红霞等[26]从测定香蕉果实发育过程中RS含量的动态变化规律开始,首先克隆了6个MaGBSSs家族成员,发现MaGBSSI-3在香蕉果实发育的中后期表达量最高[72],且与该团队2007年通过香蕉果实SSH文库筛选获得的GBSS是同一个基因[73]。同时,在香蕉A和B基因组中也存在MaGBSSI-3基因[51, 74],表明该基因进化上相对保守。番茄中异源过表达MaGBSSI-3能够促进果实RS合成[75],但MaGBSSI-3是否协同MaSSⅢ-1或MaSBE2.3共同参与RS合成仍不清楚。该团队通过异源过表达或香蕉果实薄片中瞬时过表达及瞬时沉默表达等实验,发现MaSSⅢ-1[76]和MaSBE2.3[52]主要促进果实支链淀粉合成,而未与MaGBSSI-3共同参与RS合成,进一步证明MaGBSSI-3是促进香蕉果实RS合成的关键基因。
5 香蕉抗性淀粉制备方法及其在食品加工方面的应用进展 5.1 香蕉抗性淀粉制备方法香蕉是一种优良的天然RS2来源,在食品工业中有广阔的应用前景。但由于香蕉淀粉颗粒耐热性差,当温度达到60~80 ℃时,淀粉颗粒容易在水中发生溶胀分裂,形成均匀糊状胶体溶液,极易破坏香蕉RS2结构,且香蕉中果胶、纤维素、蛋白质、色素等物质容易附着在RS2颗粒表面,通过过滤等方式无法去除。因此,学者们对香蕉果实纯化RS2工艺进行了大量研究,探索出酶解法除杂质纯化制取RS2方法,其工艺流程为:香蕉果实→去皮→护色→低温排氧打浆→酶解→分离→干燥→粉碎→ RS2,采用此法得到的RS2纯度可达99.61%~99.75%[77]。Kaur等[78]比较碱提取法和超声波法对香蕉淀粉进行预处理的影响,发现超声波预处理易对淀粉颗粒造成破坏(图 3A、C),其淀粉颗粒表面粗糙、凹陷且存在断裂情况;而碱液预处理的香蕉淀粉颗粒较为完整(图 3A、B)。

|
| A~B:酶解法制备的香蕉RS2颗粒[78];C~D:超声波提取法和碱提取法制备的香蕉淀粉颗粒[78];E:三偏磷酸钠法制备的香蕉RS4颗粒[83];F:交联磷酸化法制备的香蕉RS4颗粒[83];G:辛烯基琥珀酸酯化反应制备的香蕉RS4颗粒[39];H:柠檬酸酯化制备的香蕉RS4颗粒[84];I:水热处理法制备的香蕉RS5颗粒[46];J:物理粉碎法制备的香蕉RS1颗粒[85];K:碱提取- 高压灭菌- 冷却法制备的香蕉RS3颗粒[86];L:水热处理法制备的香蕉RS3颗粒[81];M:回生处理法制备的香蕉RS3颗粒[81];N:酶解- 水热处理法制备的香蕉RS3颗粒[82];O:酸稀释法制备的香蕉RS3颗粒[83];P:羟丙基化法制备的香蕉RS3颗粒[83];Q:氧化法制备的香蕉RS3颗粒[83];R:退火处理法制备的香蕉RS3颗粒[81] A-B: Banana resistant starch 2 (RS2) granules prepared by enzymatic hydrolysis method[78]; C-D: Banana starch granules prepared by ultrasonic microwave-assisted alkaline extraction[78]; E: Banana RS4 granules prepared by sodium trimetaphosphate method[83]; F: Banana RS4 granules prepared by cross-linking phosphorylation method[83]; G: Banana RS4 granules prepared by octenylsuccinate esterification reaction[39]; H: Banana RS4 granules prepared by citric acid esterification[84]; I: Banana RS5 granules prepared by moist heat treatment method[46]; J: Banana RS1 granules prepared by physical crushing method[85]; K: Banana RS3 granules prepared by alkali extraction-high pressure sterilization-cooling method[86]; L: Banana RS3 granules prepared by moist heat treatment method[81]; M: Banana RS3 granules prepared by retrogradation[81]; N: Banana RS3 granules prepared by enzymatic hydrolysis and moist heat treatment[82]; O: Banana RS3 granules prepared by acid dilution method[83]; P: Banana RS3 granules prepared by hydroxypropylation method[83]; Q: Banana RS3 granules prepared by oxidation method[83]; R: Banana RS3 granules prepared by annealing treatment method[81] 图 3 香蕉不同类型抗性淀粉的改性方法 Fig. 3 Modification methods of different types of resistant starch in banana |
目前关于香蕉RS制备工艺的研究多集中在RS3和RS4制备工艺优化和改进方面。其中,RS3制备方法主要有水热处理法(如湿热法、压热法、韧化法)、脱支处理法(如酶解法、酸解法)和机械辅助法(如挤压法、微波法、超声波法)。研究表明,通过优化制备工艺中淀粉乳液pH值、含水量、加热温度、贮藏条件、酸/酶种类和用量等实验条件,可促使支链淀粉链分解形成更多直链分子,从而提高RS3含量。香蕉淀粉中直链淀粉含量少,经过高压灭菌和冷却后形成的RS3含量约为6%;而香蕉淀粉经过脱支、高压灭菌和冷却等处理后制备的RS3含量高达51%[79]。唐健[80]通过正交实验筛选出香蕉RS酶解最佳条件为中温α- 淀粉酶与普鲁兰酶配比为1︰1、酶添加量为0.22%、温度59℃、pH 6.0、时间4.2 h,在此条件下香蕉RS含量为81.07%。另有研究指出,使用湿热处理、退火和双重回生等方法可对香蕉淀粉进行改性,通过比较发现RDS、SDS和RS含量受热处理的影响较大。当香蕉淀粉经热处理时,SDS含量占总淀粉含量的34.51%,RS占总淀粉含量的50%;双重回生处理将SDS含量降至14.27%,而退火对SDS含量改性效果较差[81]。使用酶解- 退火法制备香蕉RS,发现改性淀粉在傅里叶红外光谱的淀粉指纹区检测到短链双螺旋的有序度增加,且随着结晶度增加其热稳定性增加,改性后淀粉颗粒形状不规则、表面结构更加致密[82](图 3N)。
有研究将水热处理法与脱支处理法,或机械辅助法与脱支处理法等两种或多种方法联合制备RS3,以最大程度提高RS3含量[87-88]。研究表明,湿热处理法是提高香蕉淀粉热稳定性和抗剪切力的有效方法,但处理时间和温度对淀粉理化性质、流变学性质和消化率有显著影响[89]。另有研究发现,超声波处理的香蕉样品具有较高的RS3含量,且得到的RS3水解度和水解速率较低、结晶结构较好[90]。Sánchez-Rivera等[84]研究发现,柠檬酸- 水热法制备的大蕉淀粉具有较高的RS3含量(93.87%)和蒸煮稳定性,经过蒸煮后RS3颗粒呈聚集状且不易消化,可以作为营养保健食品的配方成分,具有较广阔的应用前景。
使用磷酰氯(Phosphoryl chloride, POCl3)、三聚磷酸钠(Sodium tripolyphosphate, STPP)、环氧氯丙烷(Epichlorohydrin, EPI)、三偏磷酸钠(Sodium trimetaphosphate, STMP)、辛烯基琥珀酸酐(Octenyl succinic anhydride, OSA)等化学交联试剂对淀粉进行交联、酯化等,得到化学改性淀粉,可极大拓宽RS4适用范围,使之更适合工业需求。Paramasivam等[83]比较酸稀释法、氧化法、STMP、交联磷酸化法和羟丙基化法等化学改性对天然香蕉淀粉的改性效果,发现羟丙基化法和交联磷酸化法制备的RS4含量显著高于其他方法。Bello-Flores[91]和Remya等[39]使用OSA对香蕉和大蕉的淀粉进行改性,发现RS4和SDS含量均得到显著提高。
RS5主要是淀粉中的直链淀粉与脂质/脂肪酸、蛋白质(β蛋乳球蛋白)、多酚(如原花青苷、介子酸、没食子酸)等形成的淀粉- 脂质、淀粉- 多酚、淀粉- 脂质- 蛋白质等二元或三元复合物[43-45]。淀粉- 脂质/脂肪酸复合物的制备方法主要有二甲亚砜溶剂法、热机械法、碱溶法、酶法、水热处理法[24, 92]。影响直链淀粉- 脂质复合物形成的因素有很多,包括水分含量、反应温度、加热时间、加工方法、脂肪酸种类、淀粉来源、直链淀粉含量、直链淀粉聚合度等[24],在玉米[43, 93-94]、马铃薯[95]、小麦[92]、水稻[45]、慈姑[44]等作物中已有较多研究,但与香蕉RS5淀粉相关的报道较少,Photinam等[46]比较不同脂肪酸(椰子油、葵花油和米糠油)对香蕉淀粉-脂质/脂肪酸复合物的影响发现,使用葵花油制备的香蕉淀粉- 脂质复合物的含量为51.29%,显著高于米糠油和椰子油处理(图 3I)。
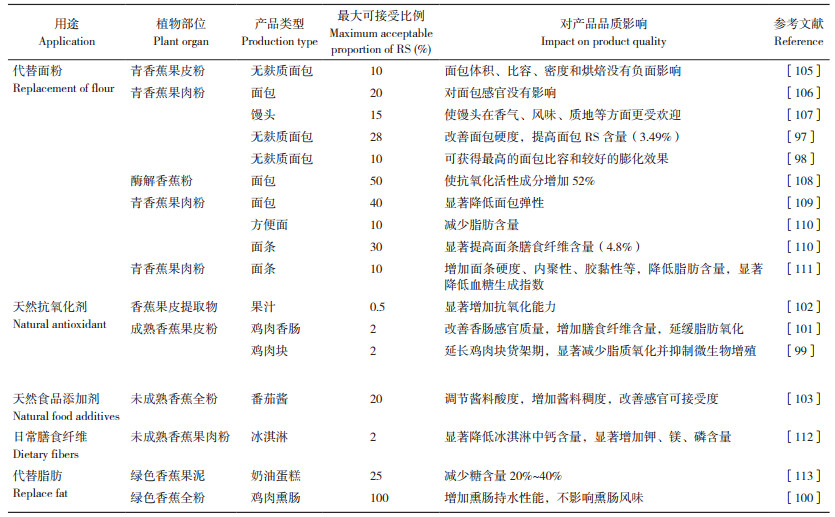
5.2 香蕉抗性淀粉在食品加工方面的应用进展青香蕉粉营养丰富,青香蕉果肉和果皮均是淀粉和膳食纤维的重要来源,在食品、饲料和工业等领域具有广阔应用前景(表 2)。众多研究认为青香蕉RS可以加入到面食和焙烤食品中以提高膳食纤维含量和营养价值,可有益于人体健康。目前,青香蕉RS相关应用研究报道主要集中在蛋糕(35%)、面包(25%)和意大利面(20%)等制作方面[96]。青香蕉粉不含麸质,可作为乳糜泻、小麦过敏和麸质不耐者适吃面包的功能性配料,满足不同消费人群的特殊需求[97-98]。青香蕉果皮粉是天然抗氧化剂,将其加入鸡肉块等产品中可以抑制脂质氧化并抑制微生物增殖,有效延长产品货架期[99]。将青香蕉RS替代脂肪添加到香肠、熏肠等食品中,对改善香肠感官质量、增加膳食纤维含量、抑制脂肪氧化等具有积极作用[100-101]。青香蕉淀粉也可作为抗氧化剂、天然食品添加剂加入到果汁和番茄酱中,调节果汁口感和酱料酸度,增加酱料稠度以获得更佳感官接受度[102-103]。有研究认为,用益生菌浸渍冻干香蕉片开发富含益生菌的零食,是香蕉RS功能性食品开发的一个研究热点[104]。
6 展望
淀粉类主食和蔬菜水果是我国居民膳食结构的重要组成部分。随着经济和社会发展,我国居民的营养状况和健康状况明显提升。然而,超重、肥胖及糖尿病、高血压、心脑血管疾病等膳食相关慢性病问题日趋严重,提高当前膳食模式中膳食纤维摄入量是坚持面向人民生命健康的重要课题。RS是一类可抵抗淀粉酶消化而不被健康人体小肠吸收的膳食纤维,引起食品、医药和农业等领域学者的极大兴趣,已成为健康食品研发的热点内容之一[64, 114]。香蕉是RS含量最高的果蔬之一,是开展RS研究和开发利用的理想材料。但是,由于香蕉果肉极易褐化,且果胶及多种酶类大量存在,导致香蕉RS制备或改性存在特殊困难。虽然目前在香蕉RS颗粒形态特征、积累特点与果实品质形成关系、制备方法及食品加工中的应用等方面取得较大进展,但与水稻等作物相比,在涉及香蕉RS合成核心基因挖掘、功能鉴定及分子育种等方面的研究明显滞后,亟需从以下2个方面开展深入研究。
6.1 融合多学科优势解析香蕉RS超微结构、关键基因功能及构建交互调控网络利用现代细胞学、结构生物学等技术,解析香蕉RS颗粒形态结构、分子结构、晶型、结晶度、糖苷键链接方式以及代谢物等结构特征,通过波谱分析、生物信息学、合成生物学等技术对香蕉RS结构进行鉴定、定性定量分析,构建超微结构模型,为制备、改性香蕉RS提供理论基础。研究香蕉RS合成与降解代谢规律,解析其遗传机制;分离鉴定控制RS合成与降解代谢的功能基因,解析基因之间、基因与蛋白之间、蛋白与蛋白之间的交互调控网络;构建激素代谢调控RS合成与降解的分子网络;分子设计育种精准培育适于糖尿病等特殊人群鲜食的RS保健型香蕉新品种,具有重要的理论意义和实际应用价值。
6.2 研发全方位多途径挖掘香蕉RS保健功能的技术探索香蕉RS功能性活性成分,全方位多途径开展鲜食蕉、煮食蕉及特色蕉果实中RS、活性成分提取、纯化、精深加工关键核心技术攻关,研发现代消费者易于接受的特色食物和保健型食品;建立涵盖香蕉产地环境、投入品管控、加工储运、适用人群、营养健康等方面的RS食品生产标准,构建多元化香蕉RS食品供给体系,满足人们健康生活需求。
| [1] |
HUANG L C, TAN H Y, ZHANG C Q, LI Q F, LIU Q Q. Starch biosynthesis in cereal endosperms: An updated review over the last decade[J]. Plant Communication, 2021(5): 100237-100259. DOI:10.1016/j.xplc.2021.100237 |
| [2] |
CAI T, SUN H B, QIAO J, ZHU L L, ZHANG F, ZHANG J, TANG Z J, WEI X L, YANG J G, YUAN Q Q, WAND W Y, YAND X, CHU H Y, WAND Q, YOU C, MA H W, SUN Y X, LI Y, LI C, JIANG H F, WANG Q H, MA Y H. Cell-free chemoenzymatic starch synthesis from carbon dioxide[J]. Science, 2021, 373(6562): 1523-1527. DOI:10.1126/science.abh4049 |
| [3] |
BALL S G, MORELL M K. From bacterial glycogen to starch: Understanding the biogenesis of the plant starch granule[J]. Annual Review of Plant Biology, 2003, 54: 207-233. DOI:10.1146/annurev.arplant.54.031902.134927 |
| [4] |
MÉRIDA A, FETTKE J. Starch granule initiation in Arabidopsis thaliana chloroplasts[J]. Plant Journal, 2021, 107(3): 688-697. DOI:10.1111/tpj.15359 |
| [5] |
ZEEMAN S C, KOSSMANN J, SMITH A M. Starch: Its metabolism, evolution, and biotechnological modification in plants[J]. Annual Review of Plant Biology, 2010, 61: 209-234. DOI:10.1146/annurevarplant-042809-112301 |
| [6] |
TEKIN T, DINCER E. Effect of resistant starch types as a prebiotic[J]. Applied Microbiology and Biotechnology, 2023, 107(2-3): 491-515. DOI:10.1007/s00253-022-12325-y |
| [7] |
ASHWAR B A, GANI A, SHAH A, WANI I A, MASOODI F A. Preparation, health benefits and applications of resistant starch—A review[J]. Starch-Stärke, 2016, 68(3-4): 287-301. DOI:10.1002/star.201500064 |
| [8] |
ENGLYST H, WIGGINS H S, CUMMINGS J H. Determination of the non-starch polysaccharides in plant foods by gas-liquid chromatography of constituent sugars as alditol acetates[J]. The Analyst, 1982, 107(1272): 307-318. DOI:10.1039/AN9820700307 |
| [9] |
ENGLYST H N, KINGMAN S M, CUMMINGS J H. Classification and measurement of nutritionally important starch fractions[J]. European Journal of Clinical Nutrition, 1992, 46(2): 33-50. |
| [10] |
WŁODARCZYK M, ŚLIŻEWSKA K. Efficiency of resistant starch and dextrins as prebiotics: A review of the existing evidence and clinical trials[J]. Nutrients, 2021, 13(11): 3808-3834. DOI:10.3390/nu13113808 |
| [11] |
JIANG F, DU C, JIANG W, WANG L, DU S K. The preparation, formation, fermentability, and applications of resistant starch[J]. International Journal of Biological Macromolecules, 2020, 150: 1155-1161. DOI:10.1016/j.ijbiomac.2019.10.124 |
| [12] |
ARAVIND N, SISSONS M, FELLOWS C M, BLAZEK J, GILBERT E P. Optimisation of resistant starch Ⅱ and Ⅲ levels in durum wheat pasta to reduce in vitro digestibility while maintaining processing and sensory characteristics[J]. Food Chemistry, 2013, 136(2): 1100-1109. DOI:10.1016/j.foodchem.2012.08.035 |
| [13] |
JUAREZ-GARCIA E, AGAMA-ACEVEDO E, SÁYAGO-AYERDI S G, RODRÍGUEZ-AMBRIZ S L, BELLO-PÉREZ L A. Composition, digestibility and application in breadmaking of banana flour[J]. Plant Foods for Human Nutrition, 2006, 61(3): 131-137. DOI:10.1007/s11130-006-0020-x |
| [14] |
FUENTES-ZARAGOZA E, SÁNCHEZ-ZAPATA E, SENDRA E. Resistant starch as prebiotic: A review[J]. Starch-Stärke, 2011, 63(7): 406-415. DOI:10.1002/star.201000099 |
| [15] |
MAZIARZ M, SHERRAARD M, JUMA S, PRASAD C, IMRHAN V, VIJAYAGOPAL P. Sensory characteristics of high-amylose maize-resistant starch in three food products[J]. Food Science & Nutrition, 2013(2): 117-124. DOI:10.1002/fsn3.15 |
| [16] |
江扬. 超高压法制备RS4型荞麦抗性淀粉及其在焙烤食品中的应用[D]. 上海: 上海应用技术大学, 2017. JIANG Y. Preparation of type 4 buckwheat starch by ultrahigh pressure treatment and its application in baked food[D]. Shanghai: Shanghai Institute of Technology, 2017. |
| [17] |
李云云. 甘薯抗性淀粉RS4的制备及其理化性质研究[D]. 广州: 华南理工大学, 2013. LI Y Y. Study on preparation and physicochemical properties of sweet potato resistant starches (RS4)[D]. Guangzhou: South China University of Technology, 2013. |
| [18] |
DING L, HUANG Q, XIANG WY, FU X, ZHANG B, WU J Y. Chemical cross-linking reduces in vitro starch digestibility of cooked potato parenchyma cells[J]. Food Hydrocolloids, 2022, 124: 107297. DOI:10.1016/j.foodhyd.2021.107297 |
| [19] |
SONG J Y, PARK J H, SHIN M. The effects of annealing and acid hydrolysis on resistant starch level and the properties of cross-linked RS4 rice starch[J]. Starch-Stärke, 2011, 63: 147-153. DOI:10.1002/star.201000097 |
| [20] |
KOO S H, LEE KY, LEE H G. Effect of cross-linking on the physicochemical and physiological properties of corn starch[J]. Food Hydrocolloids, 2010, 24(6-7): 619-625. DOI:10.1016/j.foodhyd.2010.02.009 |
| [21] |
OKUMMUS B N, TACER-CABA Z, KAHRAMAN K, NILUFERERDIL D. Resistant starch type V formation in brown lentil (Lens culinaris Medikus) starch with different lipids/fatty acids[J]. Food Chemistry, 2018, 240(1): 550-558. DOI:10.1016/j.foodchem.2017.07.157 |
| [22] |
OYEYINKA S A, SINGH S, AMONSOU E O. A review on structural, digestibility and physicochemical properties of legume starch-lipid complexes[J]. Food Chemistry, 2021, 349(1): 129165-129168. DOI:10.1016/j.foodchem.2021.129165 |
| [23] |
石少侠, 董瑶瑶, 李琪, 于修烛. 淀粉- 脂质复合物功能及营养特性研究进展[J]. 食品科学, 2020, 41(9): 238-245. DOI:10.7506/spkx1002-6630-20190427-371 SHI S X, DONG Y Y, LI Q, YU X Z. Advances in functional and nutritional properties of starch-lipid complexes[J]. Food Science, 2020, 41(9): 238-245. DOI:10.7506/spkx1002-6630-20190427-371 |
| [24] |
WANG S J, CHEN C, CAI J J, NIU B, COPELAND L, WANG S. Starch-lipid and starch-lipid-protein complexes: A comprehensive review[J]. Comprehensive Reviews in Food Science and Food Safety, 2020, 19(3): 1056-1079. DOI:10.1111/1541-4337.12550 |
| [25] |
井涛, 谢江辉, 周登博. 香蕉栽培与病虫害防治彩色图说[M]. 北京: 中国农业出版社, 2022. JING T, XIE J H, ZHOU D B. Colorful illustrations of banana cultivation and pest control[M]. Beijing: China Agriculture Press, 2022. |
| [26] |
苗红霞, 金志强, 刘伟鑫, 张建平, 孙佩光, 徐碧玉. 香蕉果实抗性淀粉变化及其与其他类型淀粉相关性分析[J]. 中国农业科学, 2013, 46(24): 5180-5187. DOI:10.3864/j.issn.0578-1752.2013.24.012 MIAO H X, JIN Z Q, LIU W X, ZHANG J P, SUN P G, XU B Y. Changes of resistant starch content and correlation with the other types of starch in banana fruit[J]. Scientia Agricultura Sinica, 2013, 46(24): 5180-5187. DOI:10.3864/j.issn.0578-1752.2013.24.012 |
| [27] |
李博, 王雪飞, 徐飞, 谭乐和, 初众, 张彦军. 菠萝蜜种子淀粉体外消化酶解动力学及血糖值分析[J]. 热带作物学报, 2018, 39(4): 774-779. DOI:10.3969/j.issn.1000-2561.2018.04.026 LI B, WANG X F, XU F, TAN L H, CHU Z, ZHANG Y J. Enzymatic hydrolysis kinetics of jackfr uit seeds starch[J]. Chinese Journal of Tropical Crops, 2018, 39(4): 774-779. DOI:10.3969/j.issn.1000-2561.2018.04.026 |
| [28] |
CHEN J, LIANG Y, LI X, CHEN L, XIE F. Supramolecular structure of jackfruit seed starch and its relationship with digestibility and physicochemical properties[J]. Carbohydrate Polymers, 2016, 150: 269-277. DOI:10.1016/j.carbpol.2016.05.030 |
| [29] |
WANG S J, COPELAND L. Effect of acid hydrolysis on starch structure and functionality: A review[J]. Critical Reviews in Food Science and Nutrition, 2015, 55(8/11): 1079-1095. DOI:10.1080/10408398.2012.684551 |
| [30] |
GOREN A, ASHLOCK D, TETLOW I J. Starch formation inside plastids of higher plants[J]. Protoplasma, 2018, 255(6): 1855-1876. DOI:10.1007/s00709-018-1259-4 |
| [31] |
钦风凌. 高直链淀粉水稻胚乳淀粉粒的发育和结构特性[D]. 扬州: 扬州大学, 2011. QIN F L. Structural properties and development of starch granule in high-amylose rice endosperm[D]. Yangzhou: Yangzhou Univeisity, 2011. |
| [32] |
TETLOW I J, EMES M J. Starch biosynthesis in the developing endosperms of grasses and cereals[J]. Agronomy, 2017, 7(4): 81-124. DOI:10.3390/agronomy7040081 |
| [33] |
SOARES C A, PERONI-OKITA F H G, CARDOSO M B, SHITAKUBO R, LAJOLO F M, CORDENUNSI B R. Plantain and banana starches: granule structural characteristics explain the differences in their starch degradation patterns[J]. Journal of Agricultural and Food Chemistry, 2011, 59(12): 6672-6681. DOI:10.1021/jf201590h |
| [34] |
苗红霞, 金志强, 孙佩光, 刘伟鑫, 魏守兴, 徐碧玉. 3个香蕉品种的果实淀粉形状与含量及风味物质比较[J]. 西北植物学报, 2014, 34(3): 560-564. DOI:10.7606/j.issn.1000-4025.2014.03.0560 MIAO H X, JIN Z Q, SUN P G, LIU W X, WEI S X, XU B Y. Comparative study on starch shape and content and fruit flavor of three banana varieties[J]. Acta Botanica Boreali-Occidentalia Sinica, 2014, 34(3): 560-564. DOI:10.7606/j.issn.1000-4025.2014.03.0560 |
| [35] |
范媛媛, 毛联凯, 夏雨, 江东文, 杨公明. 香蕉天然抗性淀粉测定方法的比较[J]. 食品科技, 2011, 36(5): 256-259. DOI:10.13684/j.cnki.spkj.2011.05.063 FAN Y Y, MAO L K, XIA Y, JIANG D W, YANG G M. Comparison of native resistant starch of green banana by using different methods[J]. Food Science and Technology, 2011, 36(5): 256-259. DOI:10.13684/j.cnki.spkj.2011.05.063 |
| [36] |
黄欢, 王绍帆, 刘敏, 韩育梅. 微波- 酶解法处理对马铃薯淀粉结构及理化性质的影响[J]. 食品科技, 2019, 44(1): 308-315. DOI:10.13684/j.cnki.spkj.2019.01.053 HUANG H, WANG S F, LIU M, HAN Y M. Effect of microwaveenzymatic hydrolysis on structure and physicochemical properties of potato starch[J]. Food Science and Technology, 2019, 44(1): 308-315. DOI:10.13684/j.cnki.spkj.2019.01.053 |
| [37] |
HUNG P V, HUONG N T M, PHI N T L, TIEN N N T. Physicochemical characteristics and in vitro digestibility of potato and cassava starches under organic acid and heat-moisture treatments[J]. International Journal of Biological Macromolecules, 2017, 95: 299-305. DOI:10.1016/j.ijbiomac.2016.11.074 |
| [38] |
HUI R, CHEN Q, FU M L, XU Q, HE G. Preparation and properties of octenyl succinic anhydride modified potato starch[J]. Food Chemistry, 2009, 114(1): 81-86. DOI:10.1016/j.foodchem.2008.09.019 |
| [39] |
REMYA R, JYOTHI A N, SREEKUMAR J. Morphological, structural and digestibility properties of RS4 enriched octenylsuccinylated sweet potato, banana and lentil starches[J]. Food Hydrocolloids, 2018, 82: 219-229. DOI:10.1016/j.foodhyd.2018.04.009 |
| [40] |
SEUNG D, SMITH A M. Starch granule initiation and morphogenesisprogress in Arabidopsis and cereals[J]. Journal of Experimental Botany, 2019, 70(3): 771-784. DOI:10.1093/jxb/ery412 |
| [41] |
朱帆, 徐广文, 姚历, 丁文平. 小麦淀粉颗粒的微观结构研究[J]. 食品科学, 2008(5): 93-96. DOI:10.3321/J.ISSN:1002-6630.2008.05.12 ZHU F, XU G W, YAO L, DING W P. Study on micro-structural properties of wheat starch granule[J]. Food Science, 2008(5): 93-96. DOI:10.3321/J.ISSN:1002-6630.2008.05.12 |
| [42] |
BAKOURI H, GUEMRA K. Etherification and cross-linking effect on physicochemical properties of Zea mays starch executed at different sequences in 1-butyl-3-methylimidazolium chloride [BMIM] Cl ionic liquid media[J]. International Journal of Biological Macromolecules, 2019, 125: 1118-1127. DOI:10.1016/j.ijbiomac.2018.12.140 |
| [43] |
AMOAKO D B, AWIKA J M. Resistant starch formation through intrahelical V-complexes between polymeric proanthocyanidins and amylase[J]. Food Chemistry, 2019, 285: 326-333. DOI:10.1016/j.foodchem.2019.01.173 |
| [44] |
RAZA H, LIANG Q, AMEER K, MA H, REN X. Dual-frequency power ultrasound effects on the complexing index, physicochemical properties, and digestion mechanism of arrowhead starch-lipid complexes[J]. UltrasonSonochem, 2022, 84: 105978-105988. DOI:10.1016/j.ultsonch.2022.105978 |
| [45] |
CHUMSRI P, PANPIPAT W, CHEONG L Z, NISOA M, CHAJAN M. Comparative evaluation of hydrothermally produced rice starch-phenolic complexes: contributions of phenolic type, plasma-activated water, and ultrasonication[J]. Foods, 2022, 11: 3826-3847. DOI:10.3390/foods11233826 |
| [46] |
PHOTINAM R, MOONGNGARM A. Effect of adding vegetable oils to starches from different botanical origins on physicochemical and digestive properties and amylose-lipid complex formation[J]. Journal of Food Science and Technology, 2023, 60(1): 393-403. DOI:10.1007/s13197-022-05626-7 |
| [47] |
刘大同, 蒋正宁, 张晓, 刘健, 杨阳, 周宾寒, 朱冬梅, 高德荣. 小麦不同灌浆速率品种籽粒淀粉合成与积累研究[J]. 植物生理学报, 2021, 57(3): 703-712. DOI:10.13592/j.cnki.ppj.2020.0378 LIU D T, JIANG Z N, ZHANG X, LIU J, YANG Y, ZHOU B H, ZHU D M, GAO D R. Studies on starch biosynthesis and accumulation in wheat varieties with different grain-filling rates[J]. Plant Physiology Journal, 2021, 57(3): 703-712. DOI:10.13592/j.cnki.ppj.2020.0378 |
| [48] |
杨思敏. 绿豆籽粒形成过程中物质积累及淀粉理化性质研究[D]. 杨凌: 西北农林科技大学, 2022. YANG S M. Study on material accumulation and starch physicochemical properties of mung bean during grain formation[D]. Yangling: Northwest A & F University, 2022. |
| [49] |
张恒栋, 黄敏, 邹应斌, 陈佳娜, 单双吕. 米粉稻籽粒直链淀粉积累特性[J]. 中国农业科学, 2021, 54(7): 1354-1364. DOI:10.3864/j.issn.0578-1752.2021.07.004 ZHANG H D, HUANG M, ZOU Y B, CHEN J N, SHAN S L. Amylose accumulation properties in the grains of noodle rice[J]. Scientia Agricultura Sinica, 2021, 54(7): 1354-1364. DOI:10.3864/j.issn.0578-1752.2021.07.004 |
| [50] |
苗红霞, 金志强, 刘伟鑫, 刘菊华, 贾彩红, 张建斌, 徐碧玉. 香蕉果实中抗性淀粉代谢与可溶性糖含量变化的相关性[J]. 植物生理学报, 2013, 49(8): 743-748. DOI:10.13592/j.cnki.ppj.2013.08.005 MIAO H X, JIN Z Q, LIU W X, LIU J H, JIA C H, ZHANG J B, XU B Y. Correlation between resistant starch metabolism and soluble sugar content of banana fruit[J]. Plant Physiology Journal, 2013, 49(8): 743-748. DOI:10.13592/j.cnki.ppj.2013.08.005 |
| [51] |
WANG Z, MIAO H X, LIU J H, XU B Y, YAO X M, XU C X, ZHAO S C, FANG X D, JIA C H, WANG J Y, ZHANG J B, LI J Y, XU Y, WANG J S, MA W H, WU Z Y, YU L L, YANG Y L, LIU C, GUO Y, SUN S L, BAURENS F C, MARTIN G, SALMON F, GARSMEUR O, YAHIAOUI N, HERVOUET C, ROUARD M, LABOUREAU N, HABAS R, RICCI S, PENG M, GUO A P, XIE J H, LI Y, DING Z H, YAN Y, TIE W W, D'HONT A, HU W, JIN Z Q. Musa balbisiana genome reveals subgenome evolution and functional divergence[J]. Nature Plants, 2019, 5(8): 810-821. DOI:10.1038/s41477-019-0452-6 |
| [52] |
MIAO H X, SUN P G, LIU Q, LIU J, JIA C, ZHAO D, XU B, JIN Z. Molecular identification of the key starch branching enzyme-encoding gene SBE2.3 and its interacting transcription factors in banana fruits[J]. Horticulture Research, 2020, 7: 101-115. DOI:10.1038/s41438-020-0325-1 |
| [53] |
JUNIOR A V, DO NASCIMENTO J R, LAJOLO F M. Molecular cloning and characterization of an alpha-amylase occurring in the pulp of ripening bananas and its expression in Pichia pastoris[J]. Journal of Agricultural and Food Chemistry, 2006, 54(21): 8222-8228. DOI:10.1021/jf060805b |
| [54] |
JOURDA C, CARDI C, GIBERT O, GIRALDO TORO A, RICCI J, MBÉGUIÉ-A-MBÉGUIÉD, YAHIAOUI N. Lineage-Specific Evolutionary histories and regulation of major starch metabolism genes during banana ripening[J]. Frontiers in Plant Science, 2016, 7: 1778-1799. DOI:10.3389/fpls.2016.01778 |
| [55] |
XIAO YY, KUANG J F, QI X N, YE Y J, WU Z X, CHEN J Y, LU W J. A comprehensive investigation of starch degradation process and identification of a transcriptional activator MabHLH6 during banana fruit ripening[J]. Plant Biotechnology Journal, 2018, 16(1): 151-164. DOI:10.1111/pbi.12756 |
| [56] |
NAKAMURA Y, STEUP M, COLLEONI C, IGLESIAS A A, BAO J, FHJITA N, TETLOW I. Molecular regulation of starch metabolism[J]. Plant Molecular Biology, 2022, 108(4-5): 289-290. DOI:10.1007/s11103-022-01253-0 |
| [57] |
KRISHNAN H B, CHEN M H. Identification of an abundant 56 kDa protein implicated in food allergy as granule-bound starch synthase[J]. Journal of Agricultural and Food Chemistry, 2013, 61(22): 5404-5409. DOI:10.1021/jf4014372 |
| [58] |
匡云波, 赖钟雄. 香蕉叶片颗粒结合性淀粉合成酶Ⅰ和可溶性淀粉合成酶Ⅲ基因的克隆与序列分析[J]. 热带作物学报, 2012, 33(1): 70-78. DOI:10.3969/j.issn.1000-2561.2012.01.016 KUANG Y B, LAI Z X. Cloning and sequence ana lysis of GBSSI and SSⅢ from the leaves in Musa spp.[J]. Chinese Journal of Tropical Crops, 2012, 33(1): 70-78. DOI:10.3969/j.issn.1000-2561.2012.01.016 |
| [59] |
ZHOU H, WANG L, LIU G, MENG X, JING Y, SHU X, KONG X, SUN J, YU H, SMITH S M, WU D, LI J. Critical roles of soluble starch synthase SSⅢa and granule-bound starch synthase waxy in synthesizing resistant starch in rice[J]. Proceedings of the National Academy of Sciences of the United States of America, 2016, 113(45): 12844-12849. DOI:10.1073/pnas.1615104113 |
| [60] |
ITOH Y, CROFTS N, ABE M, HOSAKA Y, FUJITA N. Characterization of the endosperm starch and the pleiotropic effects of biosynthetic enzymes on their properties in novel mutant rice lines with high resistant starch and amylose content[J]. Plant Science, 2017, 258: 52-60. DOI:10.1016/j.plantsci.2017.02.002 |
| [61] |
GURUNATHAN S, RAMADOSS B R, MUDILI V, SIDDAIAH C, KALAGATUR N K, BAPU J R K, MOHAN C D, ALQARAWI A A, HASHEM A, ALLAH E F A. Single nucleotide polymorphisms in starch biosynthetic genes associated with increased resistant starch concentration in rice mutant[J]. Frontier in Genetics, 2019, 10: 946-981. DOI:10.3389/fgene.2019.00946 |
| [62] |
ZHANG C, YANG Y, CHEN Z, CHEN F, PAN L, LU Y, LI Q, FAN X, SUN Z, LIU Q. Characteristics of grain physicochemical properties and the starch structure in rice carrying a mutated ALK/SSⅡa gene[J]. Journal of Agricultural and Food Chemistry, 2020, 68(47): 13950-13959. DOI:10.1021/acs.jafc.0c01471 |
| [63] |
MISHRA A, SINGH A, SHARMA M, KUMAR P, ROY J. Development of EMS-induced mutation population for amylose and resistant starch variation in bread wheat (Triticum aestivum) and identification of candidate genes responsible for amylose variation[J]. BMC Plant Biology, 2016, 16(1): 217-232. DOI:10.1186/s12870-016-0896-z |
| [64] |
SHEN L, TIAN F, CHENG Z, ZHAO Q, FENG Q, ZHAO Y, HAN B, FANG Y, LIN Y, CHEN R, WANG D, SUN W, SUN J, ZENG H, YAO N, GAO G, LUO J, XU Z, BAI S. OsMADS58 stabilizes gene regulatory circuits during rice stamen development[J]. Plants (Basel), 2022, 11(21): 2899-2918. DOI:10.3390/plants11212899 |
| [65] |
YANG D, WU L, HWANG Y S, CHEN L, HUANG N. Expression of the REB transcriptional activator in rice grains improves the yield of recombinant proteins whose genes are controlled by a Reb-responsive promoter[J]. Proceedings of the National Academy of Sciences of the United States of America, 2001, 98(20): 11438-11443. DOI:10.1073/pnas.201411298 |
| [66] |
KAWAKATSU T, YAMAMOTO M P, TOUNO S M, YASUDA H, TAKAIWA F. Compensation and interaction between RISBZ1 and RPBF during grain filling in rice[J]. Plant Journal, 2009, 59(6): 908-920. DOI:10.1111/j.1365-313X.2009.03925.x |
| [67] |
XU H, LI X, ZHANG H, WANG L, ZHU Z, GAO J, LI C, ZHU Y. High temperature inhibits the accumulation of storage materials by inducing alternative splicing of OsbZIP58 during filling stage in rice[J]. Plant Cell and Environment, 2020, 43(8): 1879-1896. DOI:10.1111/pce.13779 |
| [68] |
SCHMIDT R, SCHIPPERS J H, MIEULET D, WATANABE M, HOEFGEN R, GUIDERDONI E, MUELLER-ROEBER B. SALTRESPONSIVE ERF1 is a negative regulator of grain filling and gibberellin-mediated seedling establishment in rice[J]. Molecular Plant, 2014, 7(2): 404-421. DOI:10.1093/mp/sst131 |
| [69] |
ZHU Y, CAI X L, WANG Z Y, HONG M M. An interaction between a MYC protein and an EREBP protein is involved in transcriptional regulation of the rice Wx gene[J]. Journal of Biological Chemistry, 2003, 278(48): 47803-47811. DOI:10.1074/jbc.M302806200 |
| [70] |
BELLO B K, HOU Y, ZHAO J, JIAO G, WU Y, LI Z, WANG Y, TONG X, WANG W, YUAN W, WEI X, ZHANG J. NF-YB1-YC12-bHLH144 complex directly activates Wx to regulate grain quality in rice (Oryza sativa L.)[J]. Plant Biotechnology Journal, 2019, 17(7): 1222-1235. DOI:10.1111/pbi.13048 |
| [71] |
FENG T, WANG L, LI L, LIU Y, CHONG K, THEIßEN G, MENG Z. OsMADS14 and NF-YB1 cooperate in the direct activation of OsAGPL2 and Waxy during starch synthesis in rice endosperm[J]. New Phytologist, 2022, 234(1): 77-92. DOI:10.1111/nph.17990 |
| [72] |
MIAO H X, SUN P G, LIU W X, XU B Y, JIN Z Q. Identification of genes encoding granule-bound starch synthase involved in amylose metabolism in banana fruit[J]. PLoS ONE, 2014, 9(2): e88077. DOI:10.1371/journal.pone.0088077 |
| [73] |
XU B Y, SU W, LIU J H, WANG J B, JIN Z Q. Differentially expressed cDNAs at the early stage of banana ripening identified by suppression subtractive hybridization and cDNA microarray[J]. Planta, 2007, 226(2): 529-539. DOI:10.1007/s00425-007-0502-6 |
| [74] |
D'HONT A, DENOEUD F, AURY J M, BAURENS F C, CARREEL F, GARSMEUR O, NOEL B, BOCS S, DROC G, ROUARD M, DA SILVA C, JABBARI K, CARDI C, POULAIN J, SOUQUET M, LABADIE K, JOURDA C, LENGELLÉ J, RODIER-GOUD M, ALBERTI A, BERNARD M, CORREA M, AYYAMPALAYAM S, MCKAIN M R, LEEBENS-MACK J, BURGESS D, FREELING M, MBÉGUIÉ-AMBÉGUIÉ D, CHABANNES M, WICKER T, PANAUD O, BARBOSA J, HRIBOVA E, HESLOP-HARRISON P, HABAS R, RIBALLAN R, FRANCOIS P, POIRON C, KILIAN A, BURTHIA D, JENNY C, BAKRY F, BROWN S, GUIGNON V, KEMA G, DITA M, WAALWIJK C, JOSEPH S, DIEVART A, JAILLON O, LECLERCQ J, ARGOUT X, LYONS E, ALMEIDA A, JERIDI M, DOLEZEL J, ROUX N, RISTERUCCI A M, WEISSENBACH J, RUIZ M, GLASZMANN J C, QUÉTIER F, YAHIAOUI N, WINCKER P. The banana (Musa acuminata) genome and the evolution of monocotyledonous plants[J]. Nature, 2012, 488(7410): 213-217. DOI:10.1038/nature11241 |
| [75] |
张凯星. 香蕉果实直链淀合成关键酶基因MaGBSSI-3的功能及表达调控机制解析[D]. 海南: 海南大学, 2016. ZHANG K X. Function and regulation mechanism of MaGBSSI-3 involved in the amylose biosynthesis of banana fruit[D]. Hainan: Hainan University, 2016. |
| [76] |
MIAO H X, SUN P G, LIU Q, JIA C H, LIU J H, HU W, JIN Z Q, XU B Y. Soluble starch synthase Ⅲ-1 in amylopectin metabolism of banana fruit: characterization, expression, enzyme activity, and functional analyses[J]. Frontiers in Plant Science, 2017, 8: 454-466. DOI:10.3389/fpls.2017.00454 |
| [77] |
程燕锋, 王娟, 李尚新, 郭卫芸, 杨公明. 香蕉抗性淀粉提取工艺优化研究[J]. 中国粮油学报, 2008, 23(2): 73-76. CHEN Y F, WANG J, LI S X, GUO W Y, YANG G M. Isolation of resistant starch of banana[J]. Journal of the Chinese Cereals and Oils Association, 2008, 23(2): 73-76. |
| [78] |
KAUR B, VENKATRAO K B, PANESAR P S, CHOPRA H K, ANAL A K. Optimization of ultrasound-assisted enzymatic extraction of resistant starch from green banana peels and its structural characterization[J]. Journal of Food Science and Technology, 2022, 59(12): 4663-4672. DOI:10.1007/s13197-022-05546-6 |
| [79] |
LEHMANN U, JACOBASCH G, SCHMIEDL D. Characterization of resistant starch type Ⅲ from banana (Musa acuminata)[J]. Journal of Agricultural and Food Chemistry, 2002, 50(18): 5236-5240. DOI:10.1021/jf0203390 |
| [80] |
唐健. 香蕉抗性淀粉制备、安全性及通便和减肥功能研究[D]. 广州: 华南农业大学, 2015. TANG J. Studies on the preparation, safety, defecation function and weight loss of banana resistance starch[D]. Guangzhou: South China Agricultural University, 2015. |
| [81] |
CAHYANA Y, WIJAYA E, HALIMAH T S, MARTA H, SURYADI E, KURNIATI D. The effect of different thermal modifications on slowly digestible starch and physicochemical properties of green banana flour (Musa acuminata Colla)[J]. Food Chemistry, 2019, 274: 274-280. DOI:10.1016/j.foodchem.2018.09.004 |
| [82] |
LIAO H J, HUNG C C. Functional, thermal and structural properties of green banana flour (cv. Giant Cavendish) by de-astringency, enzymatic and hydrothermal treatments[J]. Plant Foods for Human Nutrition, 2023, 78(1): 52-60. DOI:10.1007/s11130-022-01021-x |
| [83] |
PARAMASIVAM S K, SUBRAMANIYAN P, THAYUMANAVAN S, SHIVA K N, NARAYANAN S, RAMAN P, SUBBARAYA U. Influence of chemical modifications on dynamic rheological behaviour, thermal techno-functionalities, morpho-structural characteristics and prebiotic activity of banana starches[J]. International Journal of Biological Macromolecules, 2023, 249: 126125-126141. DOI:10.1016/j.ijbiomac.2023.126125 |
| [84] |
SÁNCHEZ-RIVERA M M, MARIA N S, BELLO-PÉREZ L A, AGAMA-ACEVEDO E, ALVAREZ-RAMIREZ J. Citric acid esterification of unripe plantain flour: Physicochemical properties and starch digestibility[J]. Starch-Stärke, 2017, 25(5): 1-7. DOI:10.1002/star.201700019 |
| [85] |
EBUN-OLUWA O. The implications of sample preparation on the quantification of resistant starch type 1 and related nutritional starch fractions in plantain (Musa AAB)[J]. Food Analytical Methods, 2017, 10(6): 1909-1913. DOI:10.1007/s12161-016-0761-6 |
| [86] |
SHRESTHA S, SADIQ M B, ANAL A K. Culled banana resistant starch-soy protein isolate conjugate based emulsion enriched with astaxanthin to enhance its stability[J]. International Journal of Biological Macromolecules, 2018, 120(A): 449-459. DOI:10.1016/j.ijbiomac.2018.08.066 |
| [87] |
DUYEN T T M, HUONG N T M, PHI N T L, VAN HUNG P. Physicochemical properties and in vitro digestibility of mung-bean starches varying amylose contents under citric acid and hydrothermal treatments[J]. International Journal of Biological Macromolecules, 2020, 164: 651-658. DOI:10.1016/j.ijbiomac.2020.07.187 |
| [88] |
LIU G, ZHANG R, HUO S, LI J, WANG M, WANG W, YUAN Z, HU A, ZHENG J. Insights into the changes of structure and digestibility of microwave and heat moisture treated quinoa starch[J]. International Journal of Biological Macromolecules, 2023, 246: 125681-125704. DOI:10.1016/J.IJBIOMAC.2023.125681 |
| [89] |
CORDEIRO M J M, VELOSO C M, SANTOS L S, BONOMO R C F, CALIARI M, FONTAN R D C I. The impact of heat-moisture treatment on the properties of Musa paradisiaca L. starch and optimization of process variables[J]. Food Technology and Biotechnology, 2018, 56(4): 506-515. DOI:10.17113/ftb.56.04.18.5490 |
| [90] |
ZHANG S, ZHAO K, XU F, CHEN X, ZHU K, ZHANG Y, XIA G. Study of unripe and inferior banana flours pre-gelatinized by four different physical methods[J]. Frontiers in Nutrition, 2023, 10: 1201106-1201117. DOI:10.3389/fnut.2023.1201106 |
| [91] |
BELLO-FLORES A C, NUÑEZ-SANTIAGO C M, MARTÍNGONZALEZ M S F, BEMILLER J N, BELLO-PÉREZ L A. Preparation and characterization of octenylsuccinylated plantain starch[J]. International Journal of Biological Macromolecules, 2014, 70: 334-339. DOI:10.1016/j.ijbiomac.2014.06.061 |
| [92] |
KANG X, GAO W, WANG B, YU B, GUO L, CUI B, ABD El-ATY A M. Effect of moist and dry-heat treatment processes on the structure, physicochemical properties, and in vitro digestibility of wheat starchlauric acid complexes[J]. Food Chemistry, 2021, 351: 129303-129303. DOI:10.1016/j.foodchem.2021.129303 |
| [93] |
CAI J, CHAO C, NIU B, YU J, COPELAND L, WANG S, WANG S. Effects of debranching on the formation of maize starch-lauric acid-β-lactoglobulin complexes[J]. Journal of Agricultural and Food Chemistry, 2021, 69(32): 9086-9093. DOI:10.1021/acs.jafc.0c07230 |
| [94] |
REDDY C K, CHOI S M, LEE D J, LIM S T. Complex formation between starch and stearic acid: Effect of enzymatic debranching for starch[J]. Food Chemistry, 2017, 244: 136-142. DOI:10.1016/j.foodchem.2017.10.040 |
| [95] |
KAWAI K, TAKATO S, SASAKI T, KAJIWARA K. Complex formation, thermal properties, and in-vitro digestibility of gelatinized potato starch-fatty acid mixtures[J]. Food Hydrocolloids, 2012, 27(1): 228-234. DOI:10.1016/j.foodhyd.2011.07.003 |
| [96] |
LETICIA S K, TONIN G F, RUFFO V O. Chemical, technological and sensory quality of pasta and bakery products made with biomass and green banana flour[J]. International Journal of Food Science and Technology, 2022, 57(9): 5689-5697. DOI:10.1111/IJFS.15826 |
| [97] |
HERNÁNDEZ-AGUIRRE M A, ISLAS-HERNÁNDEZ J J, SÁNCHEZ-PARDO M E. Response surface methodology for optimization of gluten-free bread made with unripe banana flour[J]. Journal of Food Measurement and Characterization, 2019, 13(3): 1652-1660. DOI:10.1007/s11694-019-00082-y |
| [98] |
HOSOKAWA K, OHISHI K, IKEDA T, INAGAKI K. Properties of ripe banana flour and application to gluten-free breadmaking[J]. Journal of Food Processing and Preservation, 2020, 44(10): 1-31. DOI:10.1111/jfpp.14789 |
| [99] |
AKRAM T, MUSTAFA S, IIYAS K, TARIQ M R, ALI S W, ALI S, SHAFIQ M, RAO M, SAFDAR W, IFTIKHAR M, HAMEED A, MANZOOR M, AKHTAR M, UMER Z, BASHARAT Z. Supplementation of banana peel powder for the development of functional broiler nuggets[J]. PeerJ, 2022, 10: 14364. DOI:10.7717/peerj.14364 |
| [100] |
AURIEMA B E, CORREA F J, SILVA R, SOARES P T S, LIMA A L, VIDAL V A S, RAICES R S L, POLLONIO M A R, LUCHESE R H, ESMERINO E A, MATHIAS S P. Fat replacement by green banana biomass: Impact on the technological, nutritional and dynamic sensory profiling of chicken mortadella[J]. Food Research International, 2022, 152(2): 110890-110890. DOI:10.1016/j.foodres.2021.110890 |
| [101] |
ZAINI H B M, SINTANG M D B, PINDI W. The roles of banana peel powders to alter technological functionality, sensory and nutritional quality of chicken sausage[J]. Food Science & Nutrition, 2020, 8(10): 5497-5507. DOI:10.1002/fsn3.1847 |
| [102] |
ORTIZ L, DORTA E, GLORIA LOBO M, GONZÁLEZ-MENDOZA L A, DÍAZ C, GONZÁLEZ M. Use of banana (Musa acuminata Colla AAA) peel extract as an antioxidant source in orange juices[J]. Plant Foods for Human Nutrition, 2017, 72(1): 60-66. DOI:10.1007/s11130-016-0591-0 |
| [103] |
FEITOSA B F, ALCÁNTARA C M, LUCENA Y J A, OLIVEIRA E N A, CAVALCANTI M T, MARIUTTI L R B, LOPES M F. Green banana biomass (Musa spp.) as a natural food additive in artisanal tomato sauce[J]. Food Research International, 2023, 170(113021): 113037. DOI:10.1016/j.foodres.2023.113021 |
| [104] |
NIRO C M, MENDONCA G M N, PAULINO L R, SOARES V F, AZEREDO H M C. Freeze-dried banana slices carrying probiotic bacteria[J]. Foods, 2023, 12(12): 2282-2289. DOI:10.3390/foods12122282 |
| [105] |
TüRKER B, SAVLAK N, KAKC M. Effect of green banana peel flour substitution on physical characteristics of gluten-free cakes[J]. Current Research in Nutrition and Food Science Journal, 2016, 4: 197-204. DOI:10.12944/CRNFSJ.4.Special-Issue-October.25 |
| [106] |
RAJESWARI H, JAGADEESH S L, SURESH G J. Physicochemical and sensory qualities of bread fortified with banana, aonla and sapota powders[J]. Journal of Nutritional Health & Food Engineering, 2018, 8(6): 487-492. DOI:10.15406/JNHFE.2018.08.00315 |
| [107] |
LOONG C Y L, WONG C Y H. Chinese steamed bread fortified with green banana flour[J]. Food Research, 2018, 2(4): 320-330. DOI:10.26656/fr.2017.2(4).058 |
| [108] |
SOTO-MALDONADO C, CONCHA-OLMOS J, ZÚÑIGA-HANSEN M E. The effect of enzymatically treated ripe banana flour on the sensory quality and glycemic response of banana-wheat flour composite muffins[J]. Journal of Food Science and Technology, 2020, 57(10): 3621-3627. DOI:10.1007/s13197-020-04394-6 |
| [109] |
THAKAENG P, BOONLOOM T, RAWDKUEN S. Physicochemical properties of bread partially substituted with unripe green banana (Cavendish spp.) flour[J]. Molecules, 2021, 26(7): 2070-2082. DOI:10.3390/molecules26072070 |
| [110] |
VERNAZA M G, GULARTE M A, CHANG Y K. Addition of green banana flour to instant noodles: Rheological and technological properties[J]. Ciência E Agrotecnologia, 2011, 35(6): 1157-1165. DOI:10.1590/S1413-70542011000600016 |
| [111] |
LI P H, WANG C W, LU W C, CHAN Y J, WANG C R. Effect of resistant starch sources on the physical properties of dough and on the eating quality and glycemic index of salted noodles[J]. Foods, 2022, 11(6): 814-826. DOI:10.3390/foods11060814 |
| [112] |
FILIZ Y. Effects of green banana flour on the physical, chemical and sensory properties of ice cream[J]. Food Technolog y and Biotechnolog y, 2015, 53(3): 315-323. DOI:10.17113/ftb.53.03.15.3851 |
| [113] |
CAROLINE O D S N, LÍBIA D L D O, ERNANDES R D A, MOREIRA G P, SANTOS LEANDRO E D, GINANI V C, ZANDONADI R P. Textural, physical and sensory impacts of the use of green banana puree to replace fat in reduced sugar pound cakes[J]. LWT-Food Science & Technology, 2018, 89: 617-623. DOI:10.1016/j.lwt.2017.11.050 |
| [114] |
WANG Z G, WANG S L, XU Q H, KONG Q, LI F, LU L, XU Y B, WEI Y L. Synthesis and functions of resistant starch[J]. Advances in Nutrition, 2023, 14(5): 1131-1144. DOI:10.1016/j.advnut.2023.06.001 |
(责任编辑 张辉玲)
 2023, Vol. 50
2023, Vol. 50